The Kontron CG2500 is the most powerful and quietest rackmount medical edge server available today*, engineered for real-time data processing in noise-sensitive clinical environments. Its compact, robust design delivers exceptional compute performance in hospitals, labs, and imaging suites without compromising acoustic comfort or reliability.
*Based on independent testing

What is the CG2500?
How Does the CG2500 Perform Against Competition?
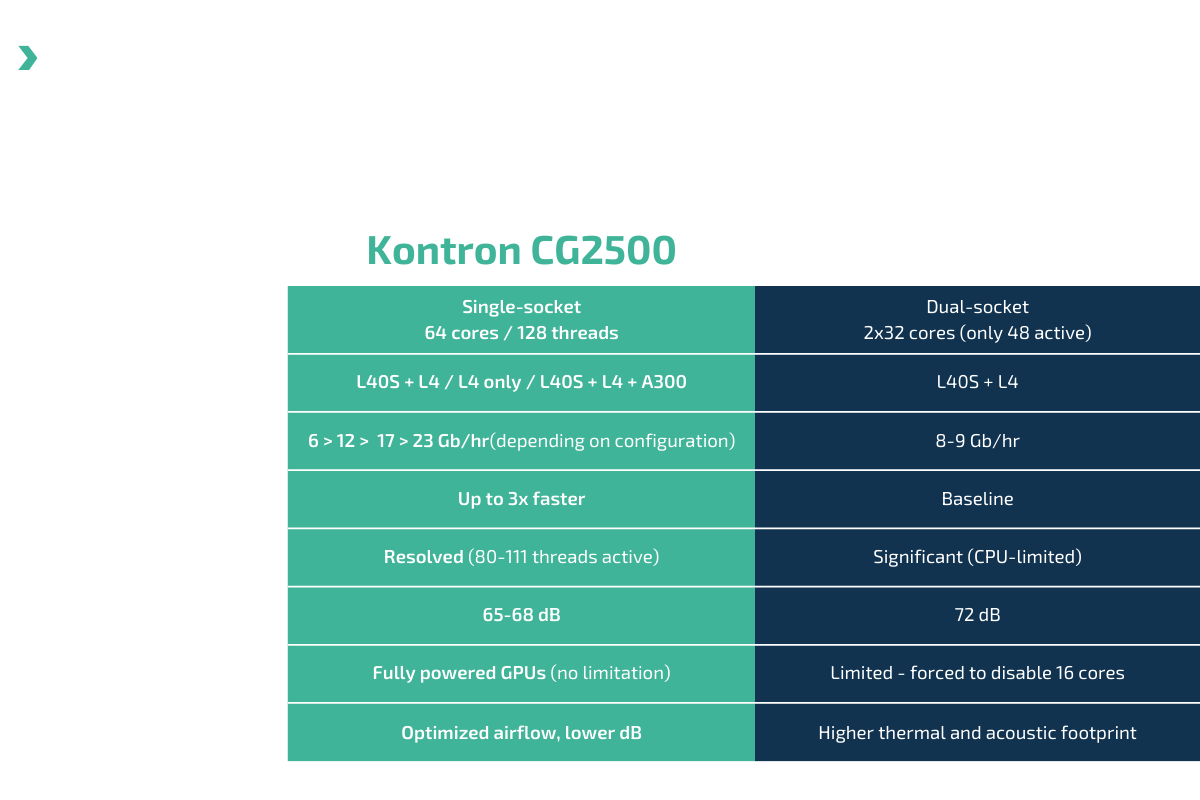
Independent testing confirms that the CG2500 is up to 3x faster throughput than typical dual-socket servers and up to 14%, 7dB quieter. It maintains full GPU power without throttling and thermal stability under heavy workloads.



Why is Quiet Operation Important in Medical Environments?
At 65–68 dB, the CG2500 runs as quietly as normal conversation, reducing distractions and improving patient comfort in imaging and lab environments. Its low noise allows in-room installation without dedicated server space.
What are the Key Advantages?
Feature | Benefit |
Quiet Operation | 65–68 dB — quietest in its class |
High Performance | Up to 3× faster throughput |
Full GPU Power | No throttling under heavy load |
Thermal Design | Cool, stable 24/7 operation |
Compact Size | 2RU, 20″ short-depth chassis |
Robust | Reliable in any environment |
Long Lifecycle | 8–10 years of platform stability |
Certified | CSA to UL 60950-1 / CSA C22.2 |
North American Manufacturing | Built in Canada; U.S. EMS available. Designed to support ISO-13485-aligned medical manufacturing workflows. |

Who Uses the CG2500?
The CG2500 is used by medical OEMs, hospitals, and research labs requiring reliable edge performance for high data workloads.
Where is the CG2500 Assembled and Tested?
Through our North American based electronics manufacturing services (EMS), Kontron delivers local design, production, and certification support for medical applications. From prototype and NPI to full-scale integration, customers benefit from faster turnaround, tariff mitigations, and in-country expertise.
Is the CG2500 Certified?
USA/CANADA | EUROPE | INTERNATIONAL |
CSA Certified (UL 60950-1, CSA C22.2 No. 60950-1-07); ISO 13485–aligned medical manufacturing support | CE mark to Low Voltage Directive 2014/35/EU and EN 62368-1 | CB report and certificate to IEC 62368-1 |
FAQ
What processors does the CG2500 support?
Both AMD EPYC™ 9004/9005 and Intel® Xeon® SP 4th/5th Gen processors, up to 128 cores.
Is the CG2500 suitable for AI or imaging workloads?
Yes. It supports PCIe Gen5 GPUs and accelerators for real-time analysis.
What certifications does it meet for medical use?
It is CSA certified to UL 60950-1 2nd Edition and CSA C22.2 with ISO 13485–aligned medical manufacturing support.
How quiet is the CG2500 compared to typical servers?
It operates at 65–68 dB vs. 72 dB or more for standard servers.
What is the product lifecycle?
The CG2500 provides an 8–10 year lifecycle for platform stability.
How powerful is the CG2500?
The CG2500 delivers up to 3× the performance of typical edge servers, enabling real-time medical imaging, genomics, and AI processing at the point of care.



